MicroRNA-29b-3p promotes 5-fluorouracil resistance via suppressing TRAF5-mediated necroptosis in human colorectal cancer
Accepted: 21 May 2021
Supplementary: 113
HTML: 13
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Authors
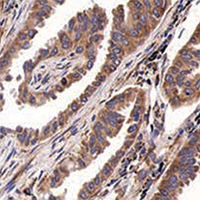
Drug resistance in colorectal cancer is a great challenge in clinic. Elucidating the deep mechanism underlying drug resistance will bring much benefit to diagnosis, therapy and prognosis in patients with colorectal cancer. In this study, miR-29b-3p was shown to be involved in resistance to 5-fluorouracil (5-FU)-induced necroptosis of colorectal cancer. Further, miR-29b-3p was shown to target a regulatory subunit of necroptosis TRAF5. Rescue of TRAF5 could reverse the effect of miR-29b-3p on 5-FU-induced necroptosis, which was consistent with the role ofnecrostatin-1 (a specific necroptosis inhibitor). Then it was demonstrated that miR-29b-3p was positively correlated with chemo-resistance in colorectal cancer while TRAF5 negatively. In conclusion, it is deduced that miR-29b-3p/TRAF5 signaling axis plays critical role in drug resistance in chemotherapy for colorectal cancer patients by regulating necroptosis. The findings in this study provide us a new target for interfere therapy in colorectal cancer.
How to Cite
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

 https://doi.org/10.4081/ejh.2021.3247
https://doi.org/10.4081/ejh.2021.3247






