Back to the future: improving storage of Golgi-stained mouse brain
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Authors
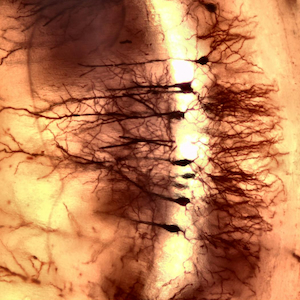
Morphological analysis of neuronal processes and their networks is a key aspect of neuroscience, with relevance from basic research to clinical practice due to the central role of neuronal development and plasticity in many neurological disorders. More than a century after its introduction, Golgi staining, a technique based on the random precipitation of metallic deposits in different neuronal subtypes, remains a highly valuable method for investigating the cellular morphology of neurons in the nervous system. Despite the wide range of protocols developed over the years, several limitations of the technique remain a matter of discussion. Among these is the need to extend sample preservation during the interval between staining and sectioning procedures without compromising the quality of the histochemical labeling. By adopting a specific processing method, the present study demonstrates that it is possible to embed murine nervous tissue following Golgi staining and to preserve the samples for extended periods prior to sectioning, while maintaining well-preserved and clearly detectable histochemical labeling across different regions and neurons of the mouse central nervous system.
CRediT authorship contribution
Fabrizio De Luca, conceptualization, methodology, investigation, data analysis, visualization, writing—original draft and review & editing.
Supporting Agencies
This work was partially funded by the NODES project, which received funding from the MUR under M4C2 1.5 of the PNRR, financed by the European Union – NextGenerationEU (Grant Agreement No. ECS00000036).Data Availability Statement
The newly generated data are reported within the manuscript.
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.









