A spectrofluorometric analysis to evaluate transcutaneous biodistribution of fluorescent nanoparticulate gel formulations
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Accepted: 17 January 2022
Authors
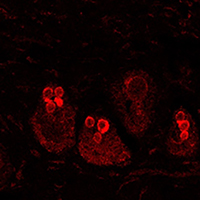
The investigation of the absorption of drug delivery systems, designed for the transport of therapeutic molecules inside the body, could be relatively simplified by the fluorophore association and tracking by means of bio-imaging techniques (i.e., optical in vivo imaging or confocal and multiphoton microscopy). However, when a fluorescence signal comes out from the skin, its specific detection can be problematic. Skin high autofluorescence can hinder the observation of administered exogenous fluorophores conjugated to drug delivery systems, making it more challenging to detect their biodistribution. In the present study, we have developed a method based on the spectrofluorometric analysis of skin samples to discriminate the fluorescent signal coming from administered fluorescent molecules from the background. Moreover, we gave a semi-quantitative evaluation of the signal intensity. Thus, we distinguished two gel formulations loading the fluorophore rhodamine B (called GEL RHO and GEL SLN-RHO). The two formulations of gels, one of which containing solid lipid nanoparticles (GEL RHO-SLN), were administered on skin explants incubated in a bioreactor, and the penetration was evaluated at different time points (2 and 6 hours). Cryostatic sections of skin samples were observed with confocal laser scanning microscopy, and a spectrofluorometric analysis was performed. Significantly higher signal intensity in the samples administered with SLN-RHO GEL, with a preferential accumulation in the hair bulbs, was found. Reaching also the deeper layers of the hair shaft after 6 hours, the solid lipid nanoparticles thickened with polymer represent a suitable drug delivery system for transcutaneous administration.
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.









